Vitek AST Kit
Regulatory & Quality
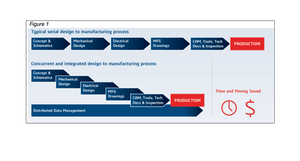
BioMérieux Vitek AST Kit Recall Given FDA Class I TreatmentBioMérieux Vitek AST Kit Recall Given FDA Class I Treatment
The recall details that the affected kits contain incorrect concentrations of Ceftriaxone antibiotic in two wells.
Sign up for the QMED & MD+DI Daily newsletter.








.png?width=300&auto=webp&quality=80&disable=upscale)





.png?width=300&auto=webp&quality=80&disable=upscale)